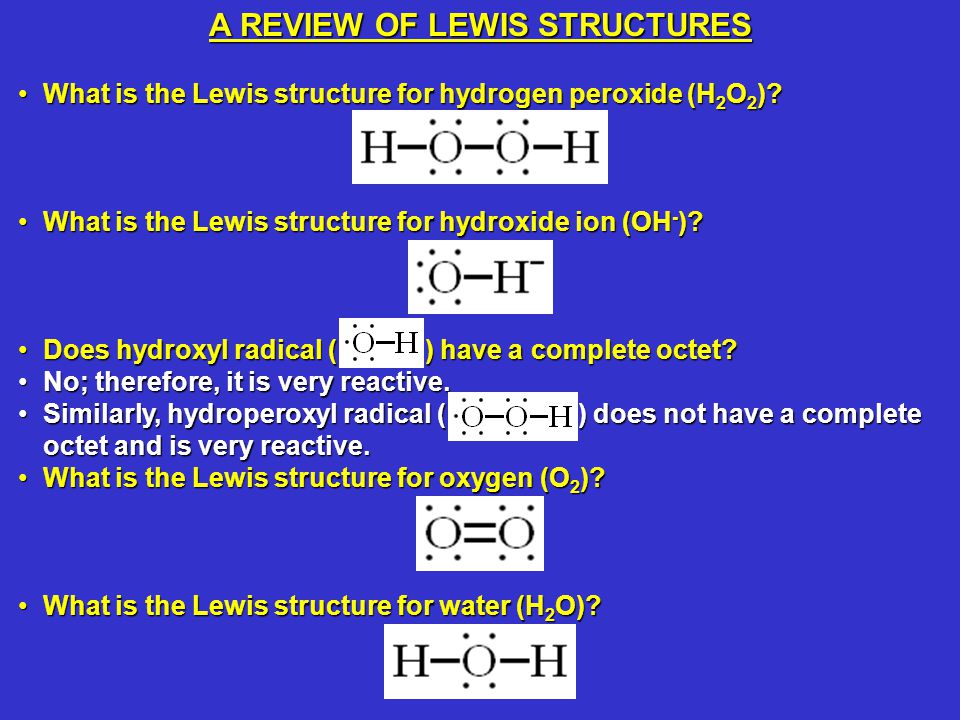
Solution for (a) Draw the Lewis structure for hydrogen peroxide, H2O2(b) What is the weakest bond in hydrogen peroxide?Put two electrons between atoms to form a chemical bond Examples CH 4, NH 3, I 2;Chemistry Hydrogen peroxide contains both a covalent and an ionic bond The bond between the oxygen atoms is covalent

Peroxide
Lewis structure for hydrogen peroxide h2o2
Lewis structure for hydrogen peroxide h2o2-Jun 01, 21 · Hydrogen Peroxide consists of 2 hydrogens and 2 oxygen atoms arranged in an open book like structure with bent OHO bonds The electronegativity of oxygen is around 344 and that of hydrogen is 22 The difference between the electronegativity of O and H atoms causes the OH bond to be polarC hydrogen peroxide, H 2O 2 (a common antiseptic) d ethylene (or ethene), C 2H 4 (used to make polyethylene) objeCtive 21 example 53 Drawing Lewis Structures from Formulas Draw a Lewis structure for each of the following formulas a phosphine, PH 3 (used to make semiconductors) b hypochlorous acid, HOCl (used to bleach textiles) c CFC



Answered A Construct A Lewis Structure For Bartleby
Volume Strength of Hydrogen Peroxide Preparation of Hydrogen Peroxide 1 From Barium Peroxide This is a method for laboratory preparation of Hydrogen Peroxide Hydrated Barium Peroxide must be used, anhydrous barium peroxide will form a protective layer and not react with the sulphuric acid The resulting H 2 O 2 is a 5% concentrate solutionHydrogen Peroxide The chemical name for H 2 O 2 is hydrogen peroxide Its Lewis structure shows us where the valence electrons are located in theUnfortunately it is not possible to provide a Lewis structure for Ca3(PO4)2 as this editor does not allow What Is The Lewis Structure For A Bicarbonate Ion?
Complete octets on outside atomsMay 06, 19 · Hydrogen peroxide is a pale blue, clear liquid, slightly more viscous than water in its pure form It is the simplest peroxide (since it is a compound with an oxygenoxygen single bond) Hydrogen peroxide has basic uses as an oxidizer, bleaching agent and antisepticThus the symmetrical Lewis structure on the left is predicted to be more stable, and it is, in fact, the structure observed experimentally We could use a lone pair on either O or Cl Hydrogen Peroxide The chemical name for H 2 O 2 is hydrogen peroxide
Jun 10, 18 · The chemical name for H2 O2 is hydrogen peroxide Its Lewis structure shows us where the valence electrons are located in the molecule, which can aid us in Count the number of electrons, add single bonds between the atoms, using two electrons per bond, arrange the remaining electrons around theToothpastes containing sodium hydrogen carbonate (sodium bicarbonate) and hydrogen peroxide are widely used Write Lewis structures for the hydrogen carbonate ion and hydrogen peroxide molecule, with resonance forms where appropriate Determine the formal charge of each element in the following (a) HCl(a) Construct a Lewis structure for hydrogen peroxide, H 2 O 2, in which each atom achieves an octet of electrons (b) How many bonding electrons are between the two oxygen atoms?(c) Do you expect the OO bond in H 2 O 2 to be longer or shorter than the OO bond in O 2?Explain



Oneclass Draw Lewis Electron Dot Structures For Ch3cl Methyl Chloride A Topical Anesthetic H2o2
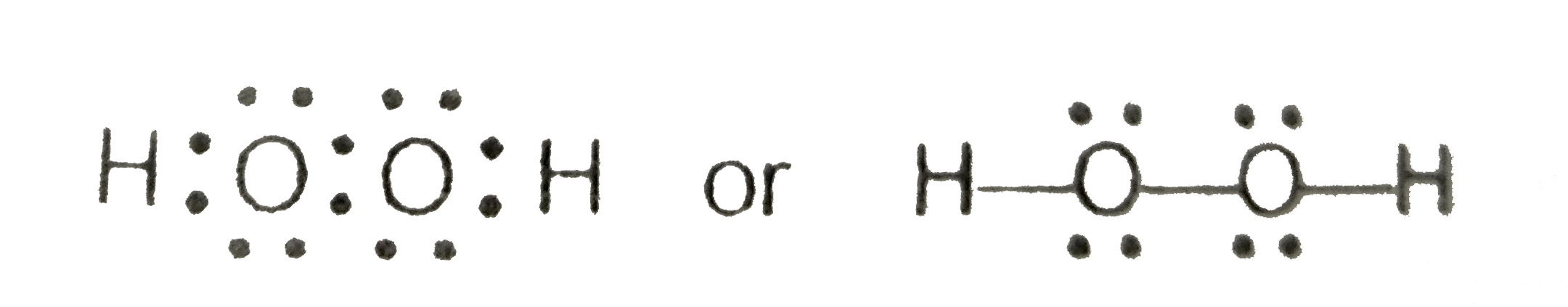


Write The Lewis Structure Of Hydrogen Peroxide
For example, we can write a Lewis structure for H2O2 as follows Indeed, H2O2, or hydrogen peroxide, exists and is often used as a disinfectant and a bleach The Lewis model also accounts for why covalent bonds are highly directional The attraction between two covalently bonded atoms is due to the sharing of one or moreToothpastes containing sodium hydrogen carbonate (sodium bicarbonate) and hydrogen peroxide are widely used(c) Hydrogen peroxide is sold commercially as an aqueous solution in brown bottles to protect it from light



What S Your Question Pricing Log Insign Up Chemistry 1 Answer 0 Watching 135 Views Titus Jacobson 28 Nov Draw The Lewis Electron Dot Structure For Hydrogen Peroxide Which Is Used To Bleach Hair Answer Watch 1 Answer 0 Watching 135 Views For



Hydrogen Peroxide Structure Clipart Lewis Structure 900x450 Png Download Pngkit
Oct 19, 18 · Toothpastes containing sodium hydrogen carbonate (sodium bicarbonate) and hydrogen peroxide are widely used Write Lewis structures for the hydrogen carbonate ion and hydrogen peroxide molecule, with resonance forms where appropriate Determine the formal charge of each element in the following (a) HCl(a) Draw the Lewis structure for hydrogen peroxide, H 2 O 2 (b) What is the weakest bond in hydrogen peroxide?(c) Hydrogen peroxide is sold commercially as an aqueous solution in brown bottles to protect it from lightCalculate the longest wavelength of light that has sufficient energy to break the weakest bond in hydrogen peroxideToothpastes containing sodium hydrogen carbonate (sodium bicarbonate) and hydrogen peroxide are widely used Write Lewis structures for the hydrogen carbonate ion and hydrogen peroxide molecule, with resonance forms where appropriate Learn this topic by watching Resonance Structures Concept Videos



Lewis H2o2 Janet Gray Coonce


Molecule Definition Examples Structures Facts Britannica
A superoxide is a compound that contains the superoxide ion, which has the chemical formula O − 2 The systematic name of the anion is dioxide(1−)The reactive oxygen ion superoxide is particularly important as the product of the oneelectron reduction of dioxygen O 2, which occurs widely in nature Molecular oxygen (dioxygen) is a diradical containing two unpaired electrons,Sep 10, 18 · It can be prepared from the reaction between benzoyl chloride and hydrogen peroxide The compound consists of two benzoyl groups that are linked by a peroxide group Benzoyl peroxide is known to have antibacterial properties and is widely used in the treatment of acne The structure of a benzoyl peroxide molecule is illustrated belowSteps for Writing Lewis Structures Find the total valence electrons for the molecule Explain How Examples H 2 S, NCl 3, OHPut the least electronegative atom in the center Note H always goes outside Examples NOCl, CF 2 Cl 2, HCN;



Lewis Structures Simple Organic Compounds Janet Gray Coonce



Answered A Construct A Lewis Structure For Bartleby
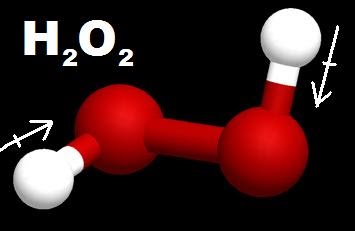
So H2O2 is a polar molecule, and that's not surprising When you go buy hydrogen peroxide in the store, it's mixed with water, and water is a polar molecule Two polar liquids will mix together So H2O2 polar molecule This is Dr B, and thanks for watchingHydrogen peroxide (H 2 O 2, HOOH) The simplest peroxide Molecular Structure of Hydrogen Peroxide Lewis structure Ball and spoke Spacefilling model Molecular model kit Related terms Hydroperoxide, hydroperoxyl radical, peracid, mCPBA,(c) Hydrogenperoxide is sold



Construct A Lewis Structure For Hydrogen Peroxide H2o2 In Which Each Atom Achieves An Octet Of Electrons


Hydrogen Peroxide Franzcalvo
Hydrogen Peroxide Structure Oxygen and Water Posted on October 18, 09 by admin Hydrogen peroxide is a naturally occuring compound formed within the cell structures of plants and animals, in the earth's atmosphere and in the waters that cover the earthHydrogen peroxide is a colorless liquid at room temperature with a bitter taste Small amounts of gaseous hydrogen peroxide occur naturally in the air Hydrogen peroxide is unstable, decomposing readily to oxygen and water with release of heat Although nonflammable, it is a powerful oxidizing agent that can cause spontaneous combustion when it comes in contact with organic materialHydrogen peroxideWrite the Lewis structures for the following, and include resonance structures where appropriate Indicate which has the strongest carbonoxygen bond CO 2;


Hydrogen Peroxide H2o2 Chemspider



Solved 19 A Student Proposed The Lewis Structure To The Chegg Com
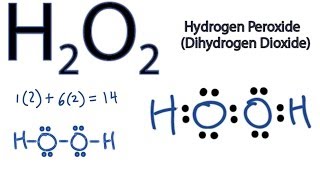
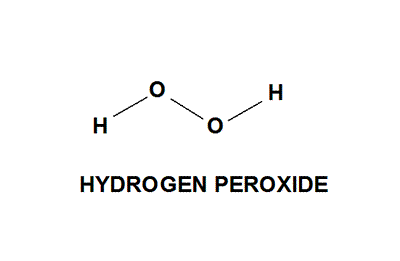
A hydrogen atom is shown as H• because of its one valence electron The structures of molecules that are held together by covalent bonds can be diagrammed by Lewis electrondot structures The hydrogen molecule is shown in the Figure 1 Figure 1 On the left is a single hydrogen atom with one electronThe Lewis structure of a water molecule then looks like O H H water Hydrogen peroxide is a different compond of hydrogen and oxygen, with chemical formula H 2O 2 Since hydrogen atoms only need a duet of electrons they are found at the outsideLewis structure of Hydrogen peroxide (H 2 O 2) contains two OH bonds and one OO bond Also, there are two lone pairs on each oxygen atom Also, there are two lone pairs on each oxygen atom Concept of number of total valence electrons of oxygen and hydrogen atoms are used to draw lewis structure of H 2 O 2


Ch 103 Percent Hydrogen Peroxide Ppt Video Online Download



Formal Charges And Resonance Chemistry For Majors
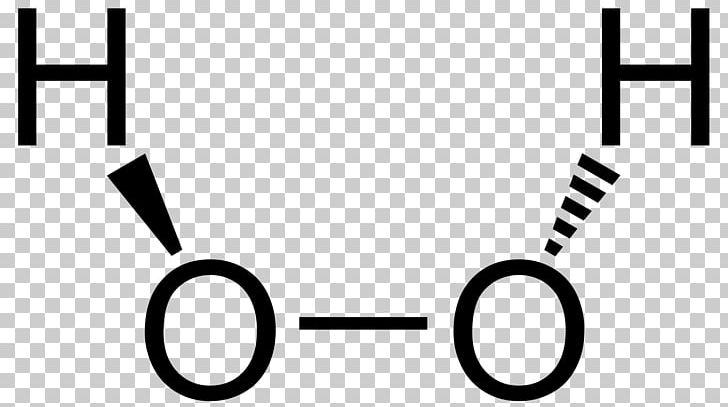
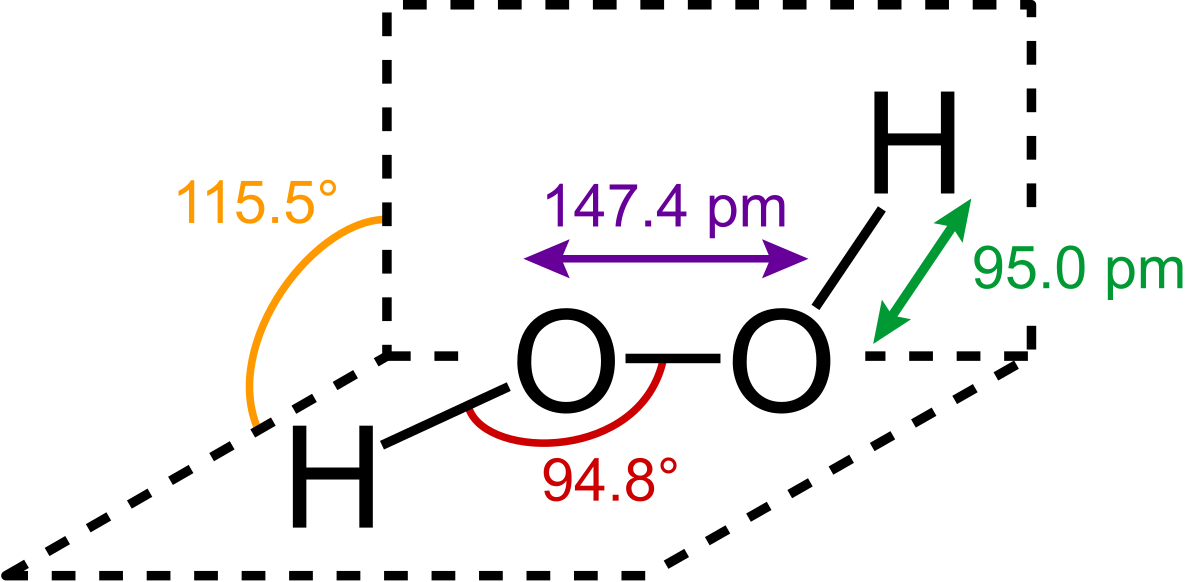
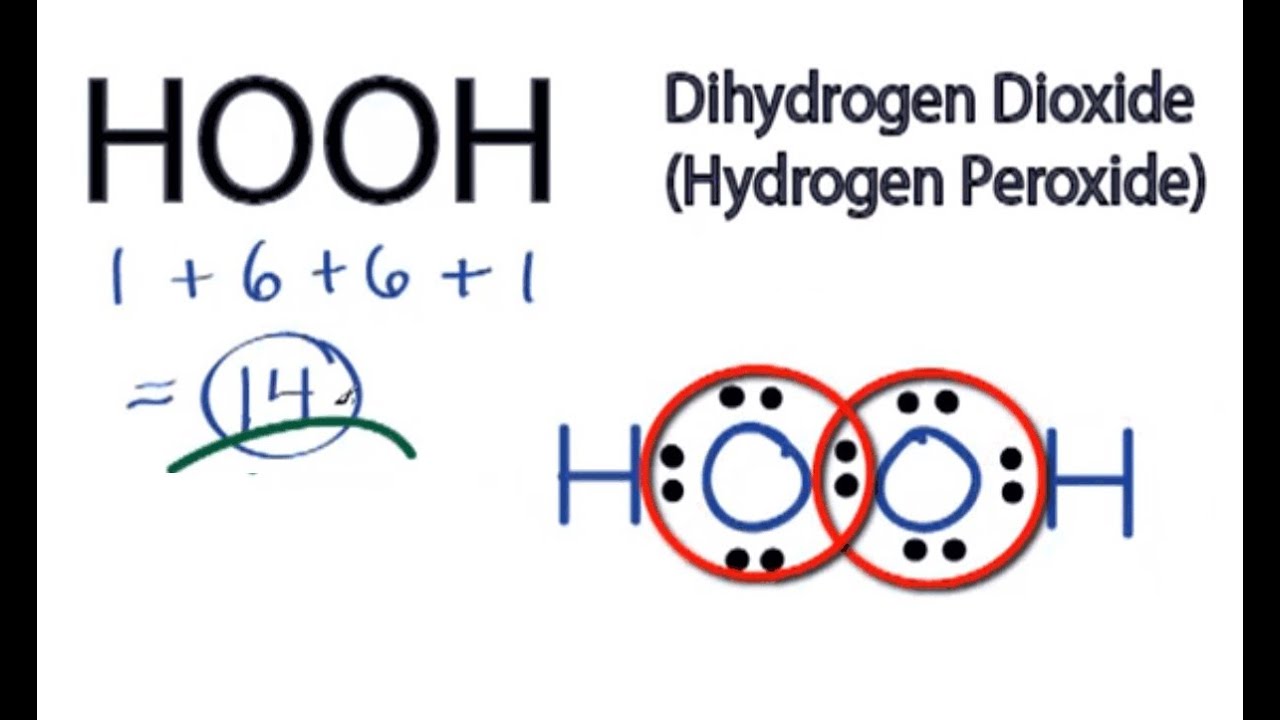
Jan 21, 19 · When drawing diagrams, hydrogen always goes on the outside So you would put down an O with an H on each side Now each hydrogen only needs 2 valence electrons, so you would draw 2 dots on each side of the O, between it and the H So you have 4 left over, so put two dots on the top, and two on the bottom So that is the Lewis dot structure Transcript This is Dr B Let's do the Lewis structure for H2O2 Hydrogen Peroxide, also called dihydrogen dioxideMay 03, 19 · The structure of hydrogen peroxide is nonplanar H 2 O 2 has an open book structure with O – O spins The dihedral angle is 111° The OO bond length is 1458 pm and the OH bond length is 9 pm (which is equal to 9 × 10 13 m)This is the Lewisdot structure of hydrogen peroxide Here you have two oxygen atoms and the overall required electrons are 6*22=14,An atom of an element is three times as heavier as the mass of an atom of carbon12 Peroxynitrite (sometimes called peroxonitrite) is an ion with the formula ONOO −It is an unstable structural isomer of



Peroxide


Hydrogen Peroxide Lewis Structure Chemistry Barium Peroxide Png Clipart 19s Angle Area Atom Barium Peroxide Free
Structure and properties In O 2 F 2, oxygen is assigned the unusual oxidation state of 1 In most of its other compounds, oxygen has an oxidation state of −2 The structure of dioxygen difluoride resembles that of hydrogen peroxide, H 2 O 2, in its large dihedral angle, which approaches 90° and C 2 symmetryCompound Source (R)6hydroxynicotine oxygen H2O > 6hydroxypseudooxynicotine hydrogen peroxide PlantCyc HYDROGENPEROXIDE, HYDROGENPEROXIDE, HYDROGENFREE Expert Solution Total number of valence electrons Group Valence Electrons H 1A 2 × 1 e– = 2 e– O 6A 2 × 6 e– = 12 e– Total 14 valence e– Bonding preferences • Hydrogen Never goes in the center and forms only one (1) bond • Oxygen Prefers to form two (2) bonds
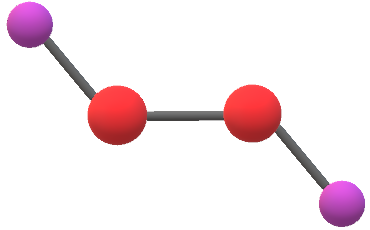


H2o2 Lewis Structure Hydrogen Peroxide Molecular Geometry Polarity



How Many Bonding Electrons Are Between The Clutch Prep
Let's do the Lewis structure for H2O2 Hydrogen Peroxide, also called dihydrogen dioxide On the periodic table, Hydrogen's in group 1 so it has 1 valence electron, but we have two of them, so we need to multiply by 2 Oxygen, group 6 or 16, we have two of those, so let's multiply that by 2 as well for a total of 14 valence electronsNov 11, 17 · The Lewis structure shows an arrangement of how the elements are connected to each other within a compound using electron dots or bonds They follow the octet rule, wherein each element must be surrounded by 8 electrons Each single bond consists of 2 electrons An exception to this rule is the hydrogen atomHydrogen peroxide dosedependently increased the intracellular ROS generation, which was significantly repressed by HW, both in the cytoplasm and nuclei LIVE/DEAD staining and our original cell viability dyeextraction assay showed that HW significantly protected HGF cells from hydrogen peroxideinduced cell death
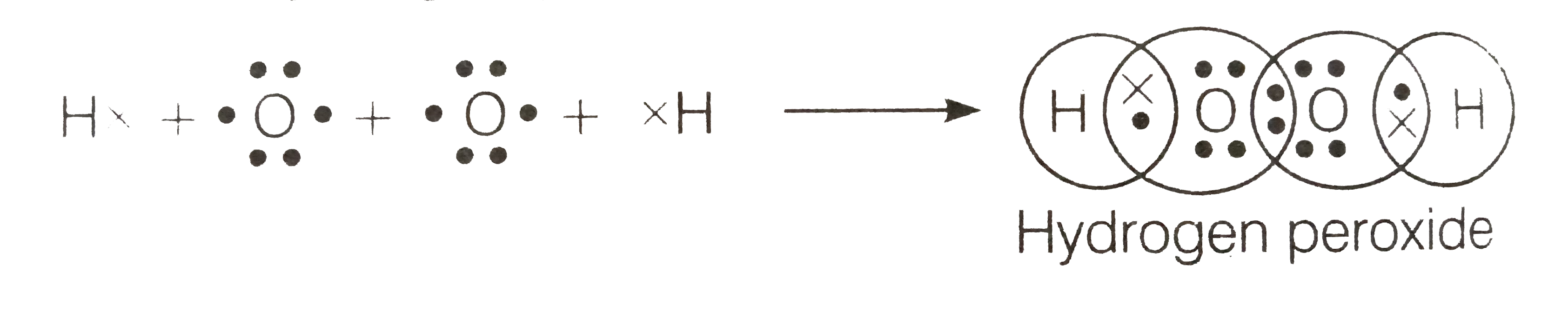


Write The Lewis Structure Of Hydrogen Peroxide



Peroxide
8 rows · The total lone pair present in the H2O2 lewis dot structure is 4 H2O2 molecular geometry is(a) Draw the Lewis structure for hydrogen peroxide, $\mathrm{H}_{2} \mathrm{O}_{2}$ (b) What is the weakest bond in hydrogen peroxide?Write the Lewis structure for the following compound Hydrogen peroxide ({eq}H_2O_2 {/eq})



What Is The Lewis Structure For H2o2 Study Com
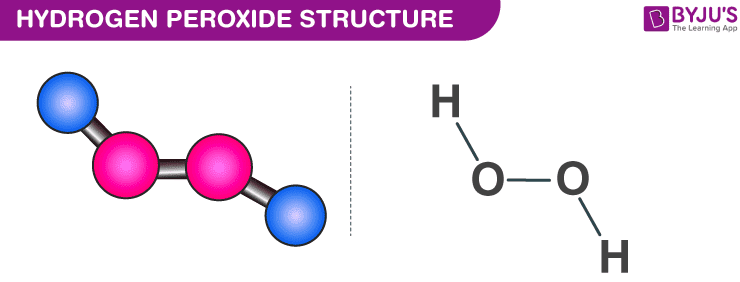


Hydrogen Peroxide H2o2 Structure Preparation Properties Uses
Chemistry Ocoh o Is Hydrogen Peroxide A Covalent Bond?Feb 25, 11 · Hydrogen peroxide has a bent shape and is a polar molecule It may seem odd that hydrogen peroxide has a bent shape When the Lewis structure is drawn it looks similar to this HOOH , with two unshared pairs on each of the oxygen This may appear to be a linear shaped molecule, but after further research this is not the caseHydrogen peroxide, H2O2, the oxygen atoms are in the center (H–O–O–H) 7 In drawing Lewis structures for relatively small molecules and polyatomic ions, the structures tend to be more stable when they are compact and symmetrical rather than extended chains of atoms EXAMPLE Write the Lewis structure for CH2O where carbon is the central
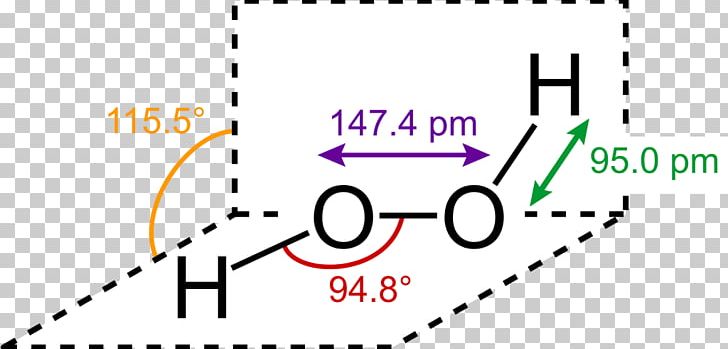


Lewis Structure Hydrogen Peroxide Molecule Structural Formula Png Clipart Angle Brand Chemical Bond Chemical Compound Chemical



Write The Lewis Structure Of Hydrogen Peroxide
C) Hydrogen peroxide is sold commercially as an aqueous solution in brown bottles to protect it from light Calculate the longest wavelength of light that has sufficient energy to break the weakest bond in hydrogen peroxideWrite the Lewis structures for the following, and include resonance structures where appropriate Indicate which has the strongest carbonoxygen bond (a) CO 2 (b) CO Toothpastes containing sodium hydrogen carbonate (sodium bicarbonate) and hydrogen peroxide are widely usedNov , 17 · a) Draw Lewis structure for hydrogen peroxide, H2O2 b) What is the weakest bond in hydrogen peroxide?



Construct A Lewis Structure For Hydrogen Peroxide H 2o 2 In Which Each Atom Achieves An Octet Of Electrons Study Com



H2o2 Lewis Structure How To Draw The Dot Structure For H2o2 Molecular Geometry Molecular Shapes Intermolecular Force
O 2 2(peroxide ion) Lewis Structure O 2 2(peroxide ion) anion contains only two oxygen atoms Peroxide anion has 2 charge In O 2 2lewis structure, each oxygen atom has 1 charge and three lone pairs Both oxygen atoms are joint through a single bond In this tutorial, we are going to draw the lewis structure of O 2 2ion step by stepDraw the lewis structure of Hydrogen Peroxide Circle one bond and determine Bond polarity including electronegativity values for the two atoms and the EN calculation Also determine the type of bond including both sigman and pi bonds Also circle one atom in the structure and determine the formal charge of it
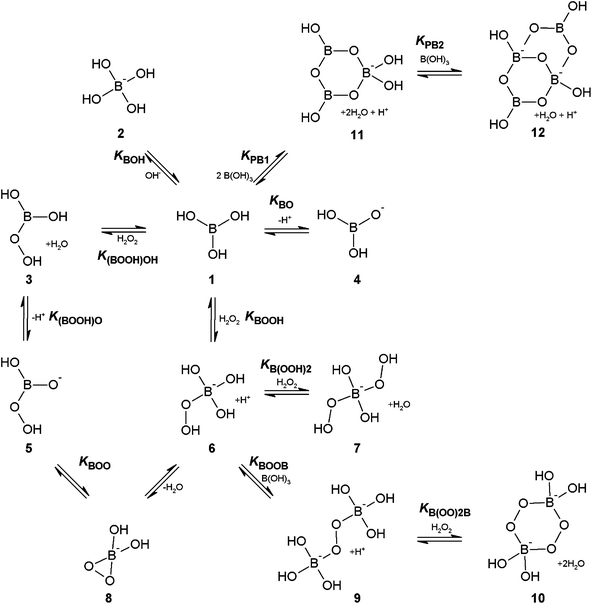


A Kinetic And Theoretical Study Of The Borate Catalysed Reactions Of Hydrogen Peroxide The Role Of Dioxaborirane As The Catalytic Intermediate For A Organic Biomolecular Chemistry Rsc Publishing Doi 10 1039 C2obf
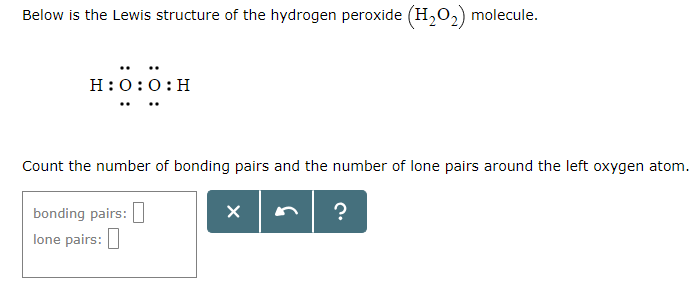


Solved Below Is The Lewis Structure Of The Hydrogen Perox Chegg Com
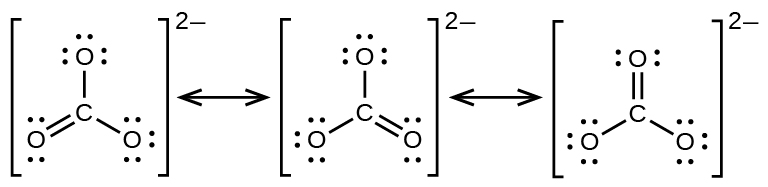


Formal Charges And Resonance Chemistry



1 E Structure And Bonding In Organic Molecules Exercises Chemistry Libretexts
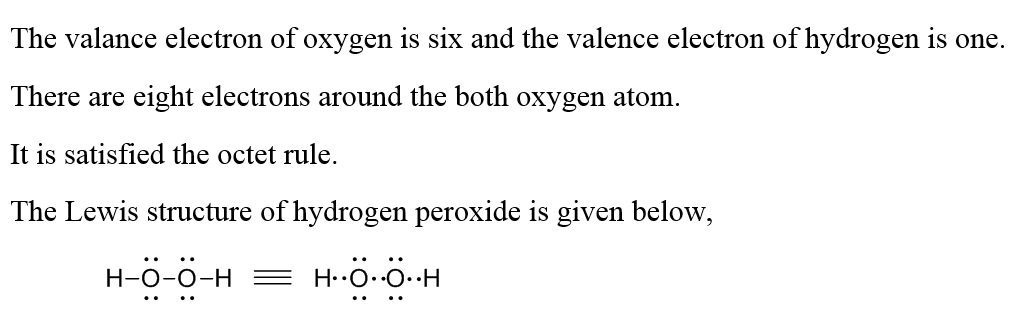


Answered A Construct A Lewis Structure For Bartleby
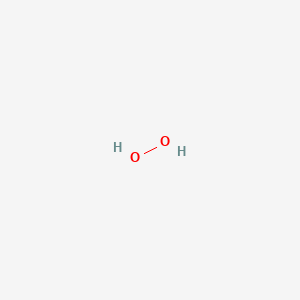


Hydrogen Peroxide H2o2 Pubchem



Reduction Of O2 To H2o And Its Free Radical Intermediates A Lewis Download Scientific Diagram
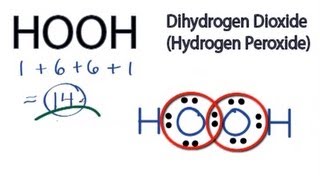


Hooh Lewis Structure How To Draw The Lewis Structure For Hydrogen Peroxide Youtube


Sodium Peroxide Na2o2 Pubchem
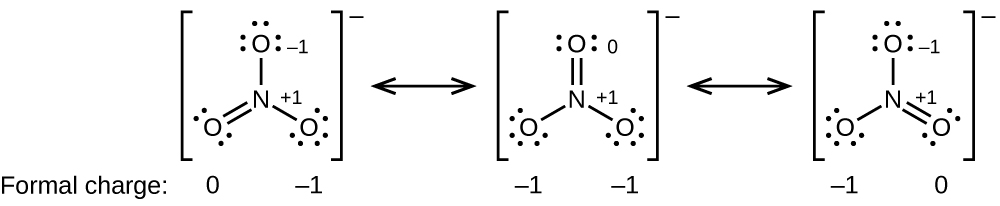


Formal Charges And Resonance Chemistry 2e
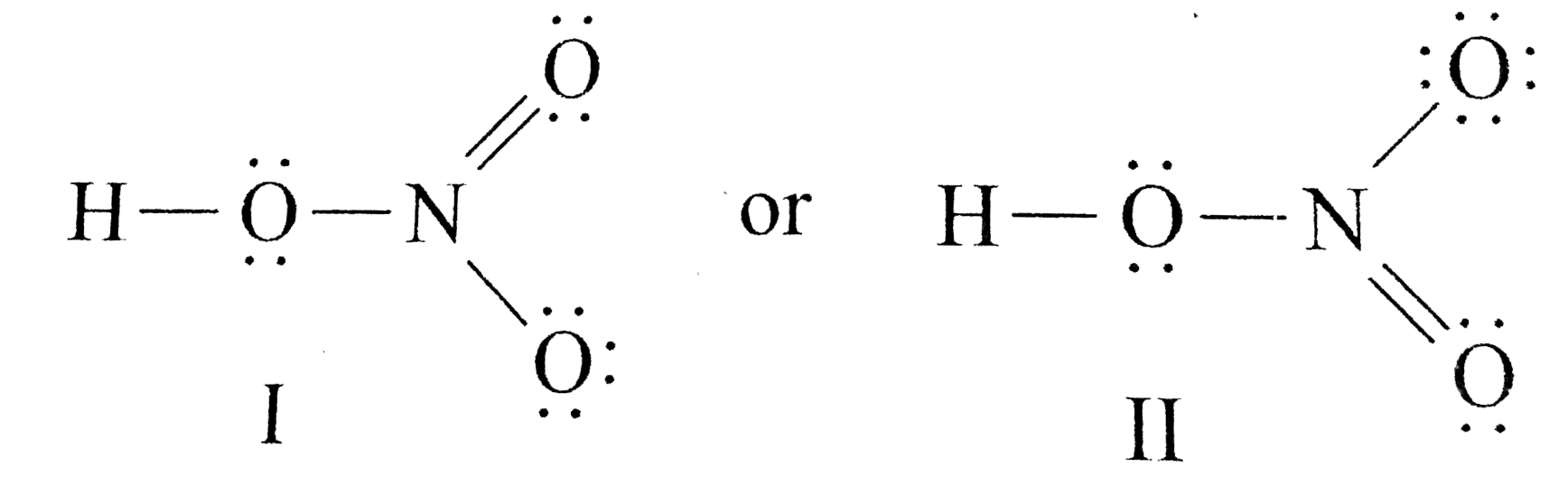


Draw The Lewis Structure Of Nitric Acid Hno 3



Hydrogen Peroxide Structure Properties Uses With Questions Videos
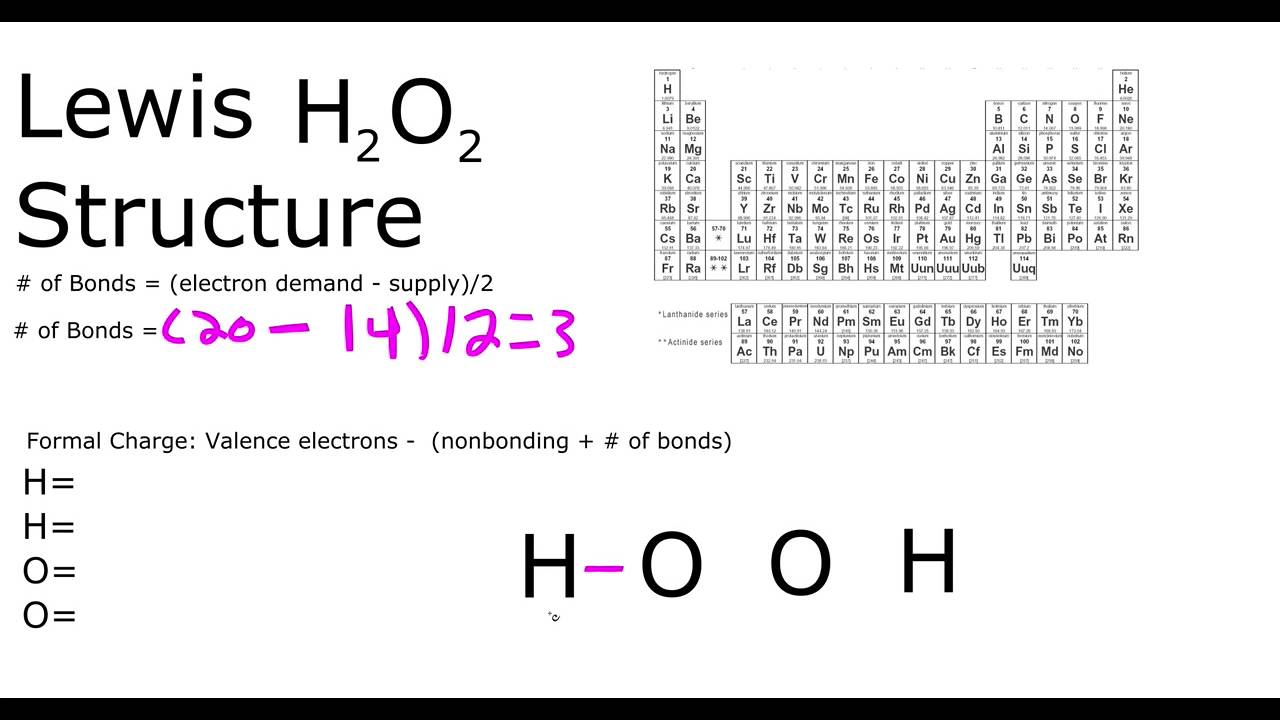


H2o2 Lewis Structure Youtube
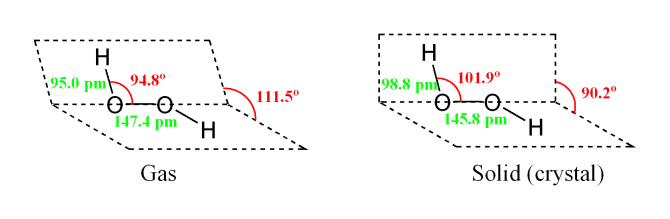


File H2o2 Structure Png Wikimedia Commons
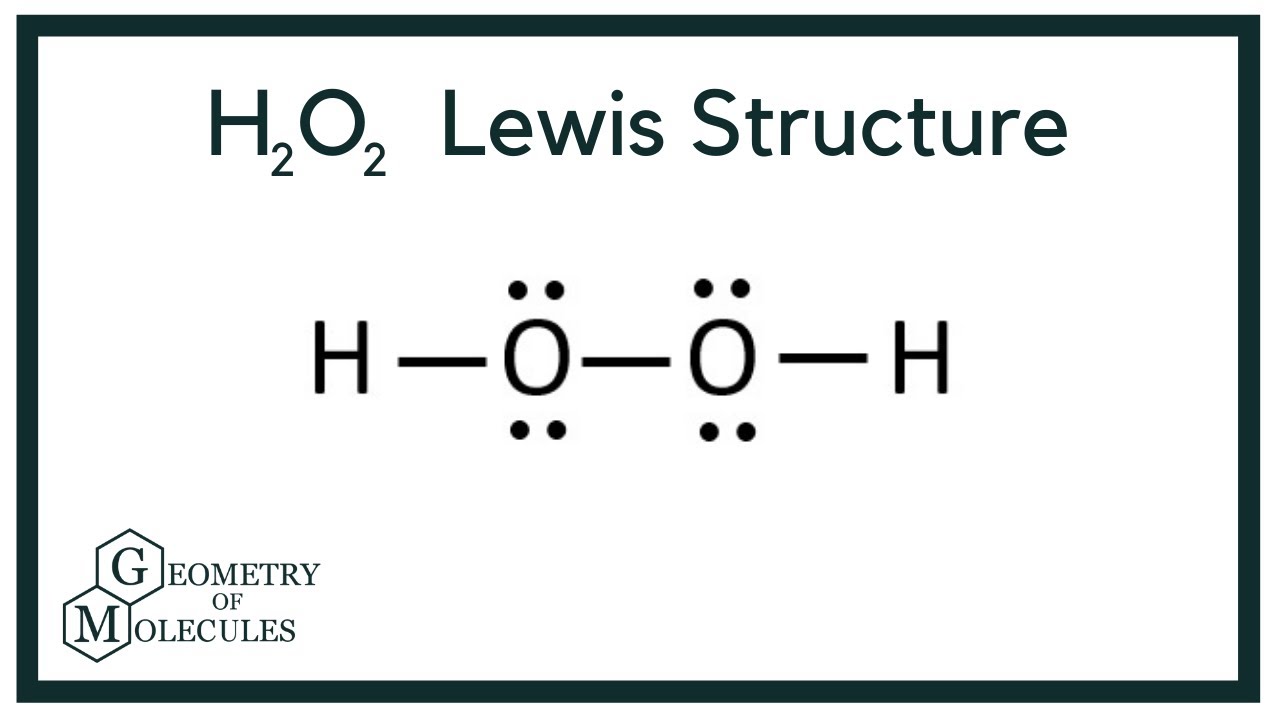


H2o2 Lewis Structure Hydrogen Peroxide Youtube



Write The Lewis Structure Of Hydrogen Peroxide
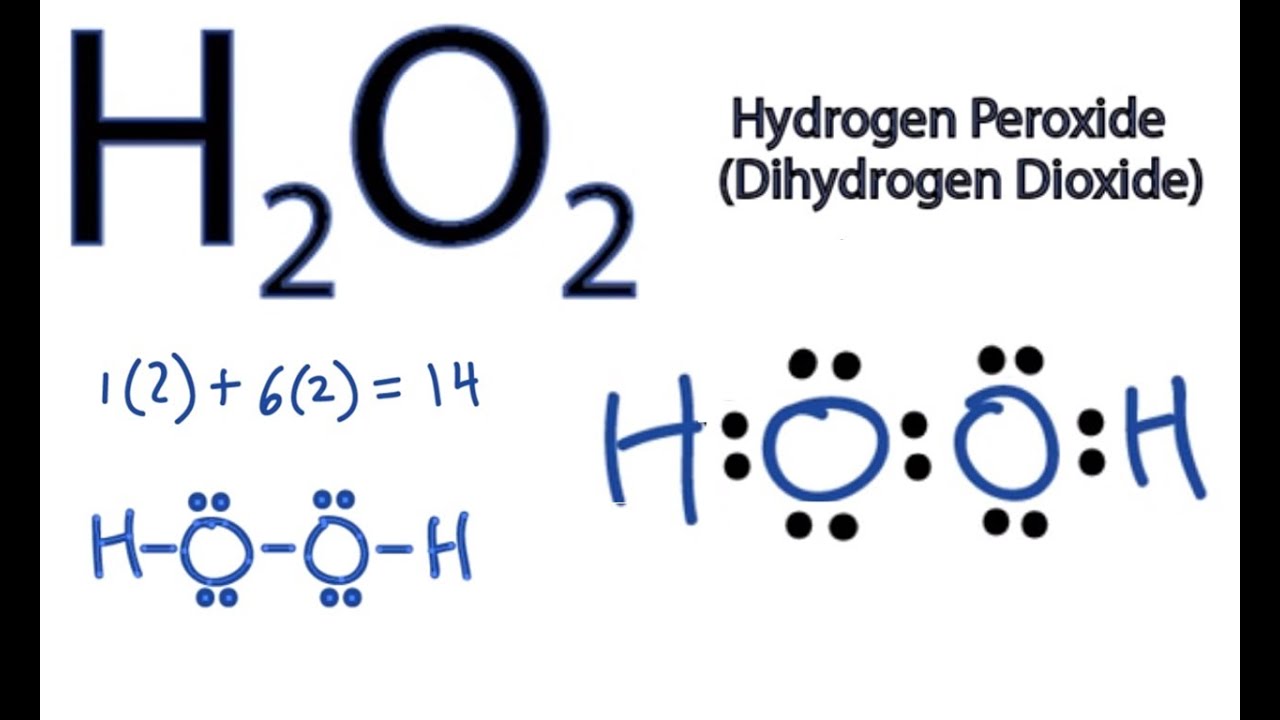


Is H2o2 Polar Or Nonpolar Youtube



Hydrogen Peroxide H2o2 Molecule Chemical Structure Hooh Is Royalty Free Cliparts Vectors And Stock Illustration Image


Hydrogen Peroxide



Lewis Structure Hydrogen Peroxide Molecule Structural Formula Laughing Gas Structure Angle Text Png Pngegg


Solved Toothpastes Containing Sodium Hydrogen Carbonate Sodium Bicarbonate And Hydrogen Peroxide Are Widely Used Write Lewis Structures For The Course Hero
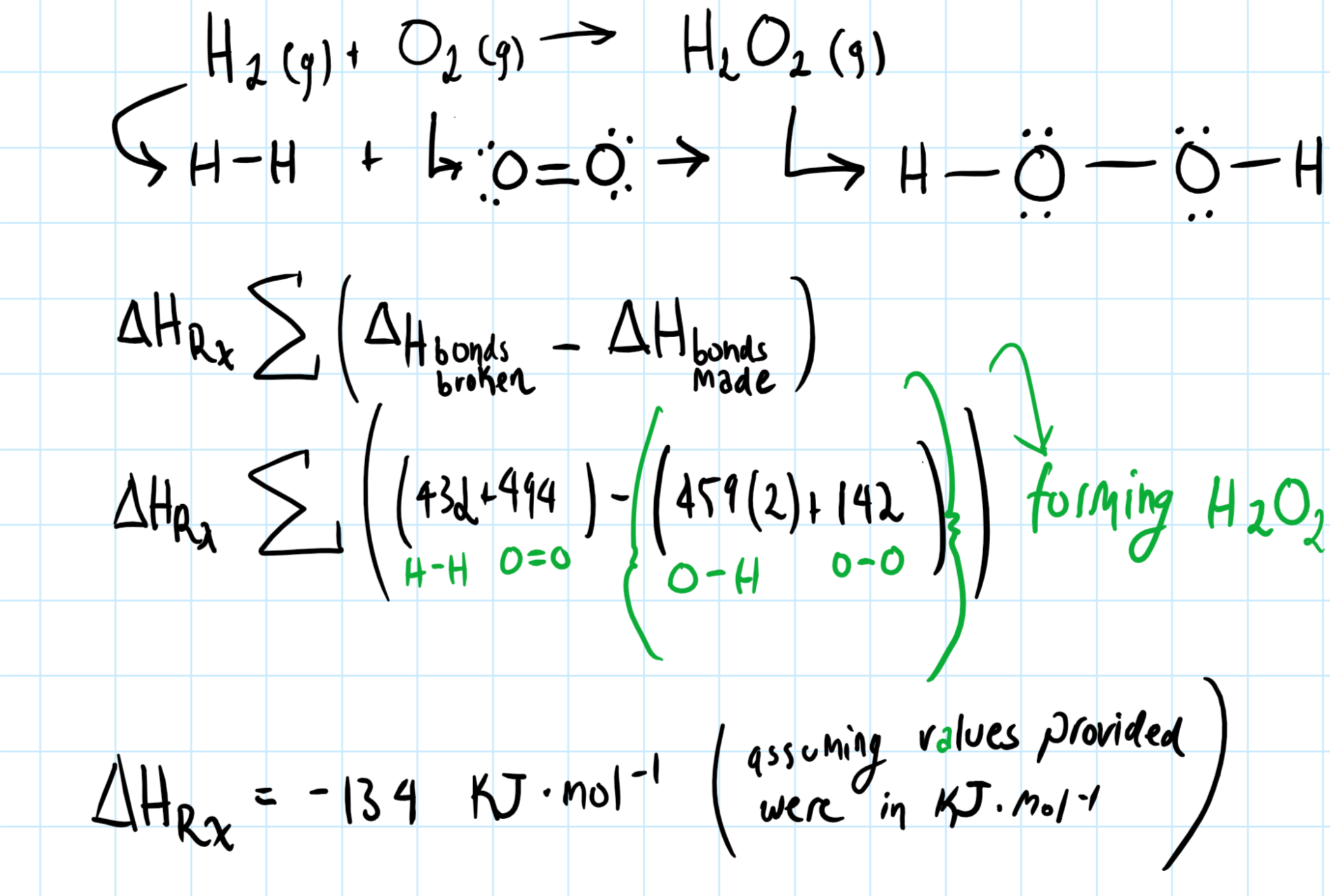


How Do You Determine The Heat Of Formation Of Hydrogen Peroxide From The Given Bond Energies H 2 G O 2 G H 2o 2 G Bond Energies H H 432 O2 494 O H 459 O O 142 Socratic



Lewis Electron Dot Structures Chemistry For Non Majors


Making Molecules Lewis Structures And Molecular Geometries Annenberg Learner


Peroxide Wikipedia


Vanadium


Illustrated Glossary Of Organic Chemistry Hydrogen Peroxide H2o2 Hooh


Magnesium Peroxide Wikipedia


7 3 Lewis Symbols And Structures Chemistry



Draw The Lewis Structure For Hydrogen Pero Clutch Prep



Hydrazine Nh2 Nh2 Hydrogen Peroxide H Clutch Prep



4 E Exercise Chemistry Libretexts



Write The Lewis Structure Of Hydrogen Peroxide



The Lewis Structure For H2o2 Novocom Top


H2o2 Lewis Structure How To Draw The Dot Structure For H2o2 Chemical Bonding Success In Chemistry


How To Draw The Lewis Structure Of 2 H2o On Paper I Need It For The Exam Quora
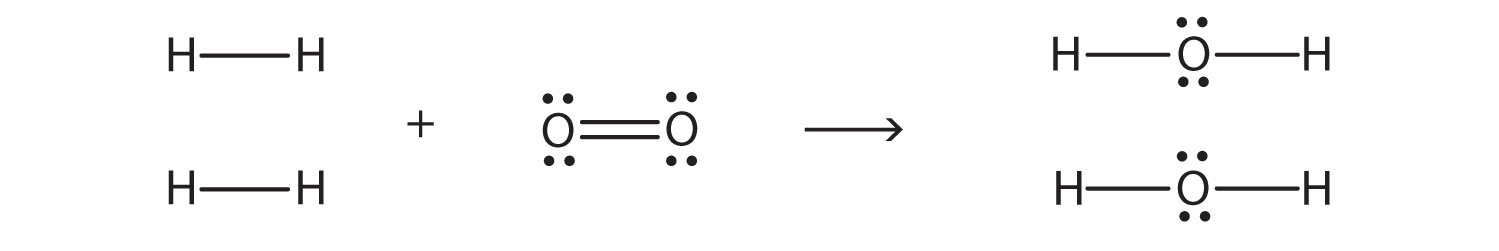


Chapter 8 Chemical Bonds Che 105 110 Introduction To Chemistry Textbook Libguides At Hostos Community College Library



Is H2o2 Polar Or Non Polar Quora



Hydrogen Peroxide Molecule And Its Lewis Dot Electron Dot Structure H2o2 Formal Science Chemistry Essay Essaysusa Com



Chapter 10 The Shapes Of Molecules Ppt Video Online Download
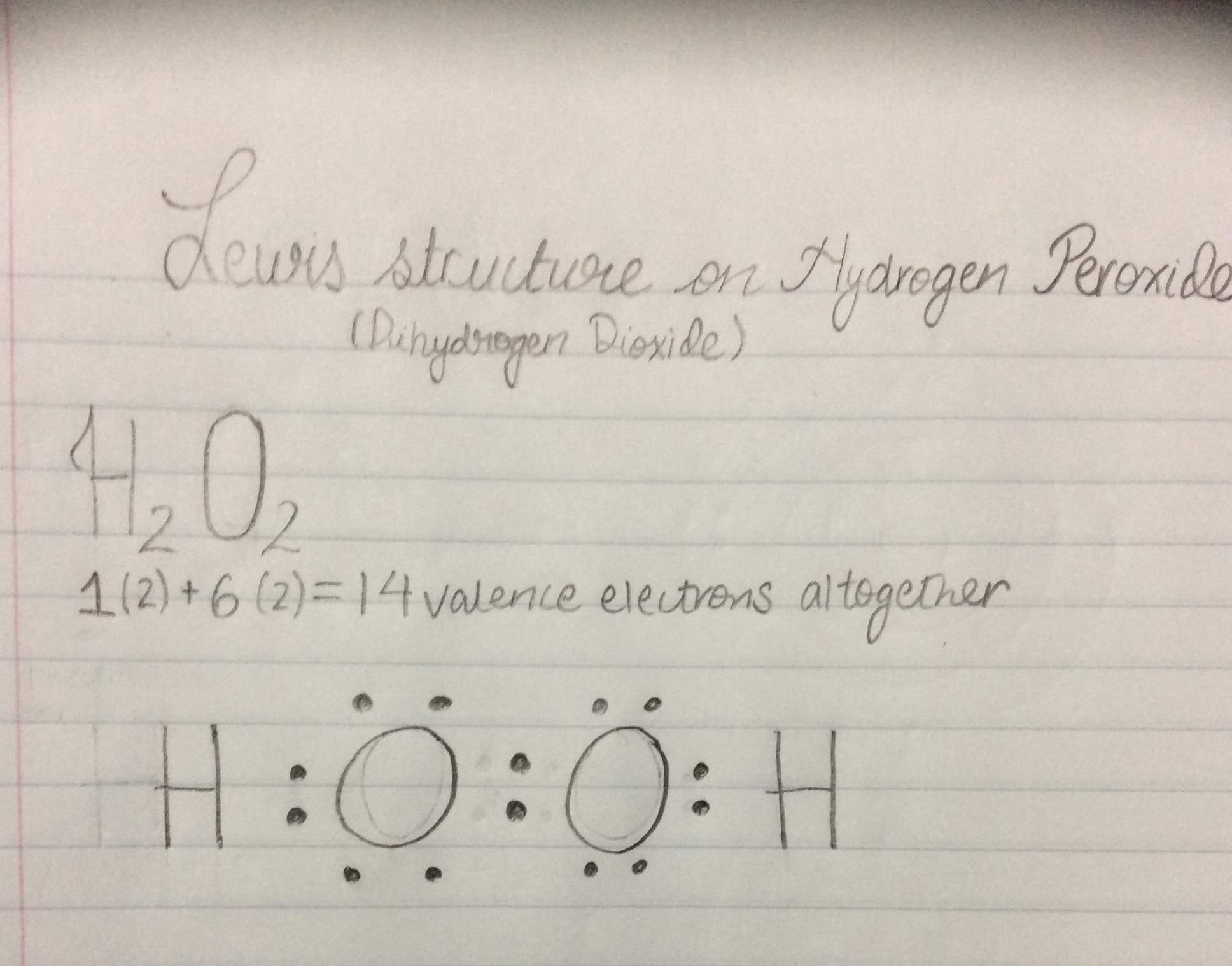


Hazards Of Hydrogen Peroxide In Hair Dye


Chem4kids Com Hydrogen Orbitals And Compounds


Hydrogen Peroxide Features And Uses



Food Grade Hydrogen Peroxide Food Grade Hydrogen Peroxide Hydrogen Peroxide Food Grade



Hydrogen Peroxide Lewis Dot Novocom Top


Hooh Lewis Structure How To Draw The Lewis Structure For Hydrogen Peroxide Youtube



Is H2o2 Polar Or Nonpolar Techiescientist



Draw The Lewis Structure For Hydrogen Pero Clutch Prep



Between The Two Structures See Image Which Is The Correct Lewis Structure For Hydrogen Peroxide Inorganic Chemistry Symmetry Of Molecules Chemistryhelp



Solved H2 Below Is The Lewis Structure Of The Hydroge Chegg Com


Hydrogenperoxide 1 Ho2 Pubchem


How To Determine The Lewis Structure For H2c2o4 Quora



Lewis Structures Simple Organic Compounds Janet Gray Coonce



Hydrogen Peroxide H2o2 Lewis Structure Novocom Top



Lewis Structure High Res Stock Images Shutterstock
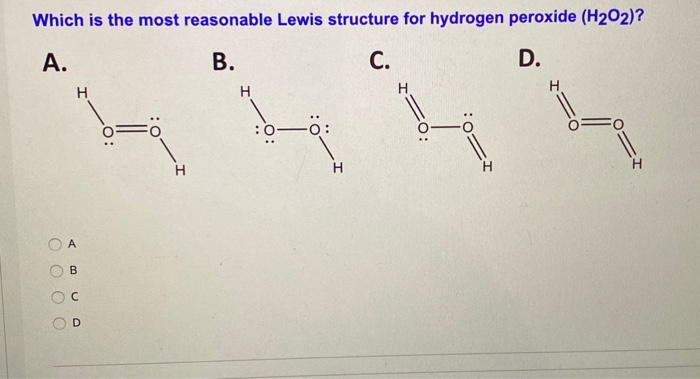


Solved Which Is The Most Reasonable Lewis Structure For H Chegg Com



Write The Lewis Structure Of Hydrogen Peroxide Youtube


Free Radicals And Reactive Oxygen



8n8j2g6swcutwm
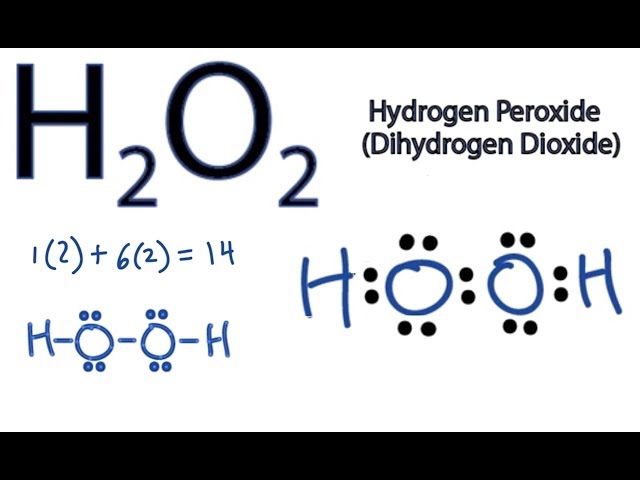


H2o2 Lewis Structure How To Draw The Dot Structure For H2o2 Youtube


Hydrogen Peroxide Molecule Of The Month September 06 Html Version



Chemistry Question Socratic
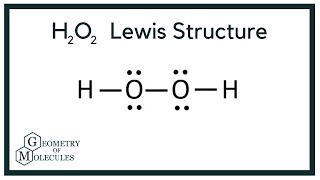


H2o2 Lewis Structure Hydrogen Peroxide Youtube


Carbamide Peroxide Ch6n2o3 Pubchem


Chem4kids Com Hydrogen Orbitals And Compounds
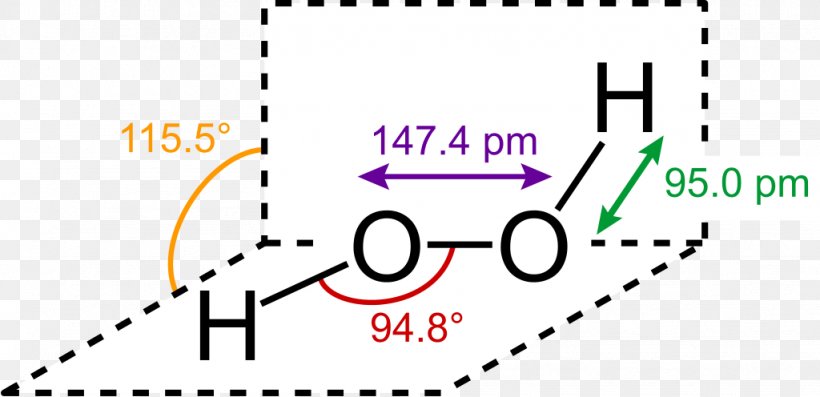


Lewis Structure Hydrogen Peroxide Molecule Structural Formula Png 1024x496px Lewis Structure Area Brand Chemical Bond Chemical



Hydrogen Peroxide Molecule Chemical Compound Lewis Structure Decomposition Hydrogen Catalysis Png Pngegg



Write The Lewis Structure Of Hydrogen Peroxide
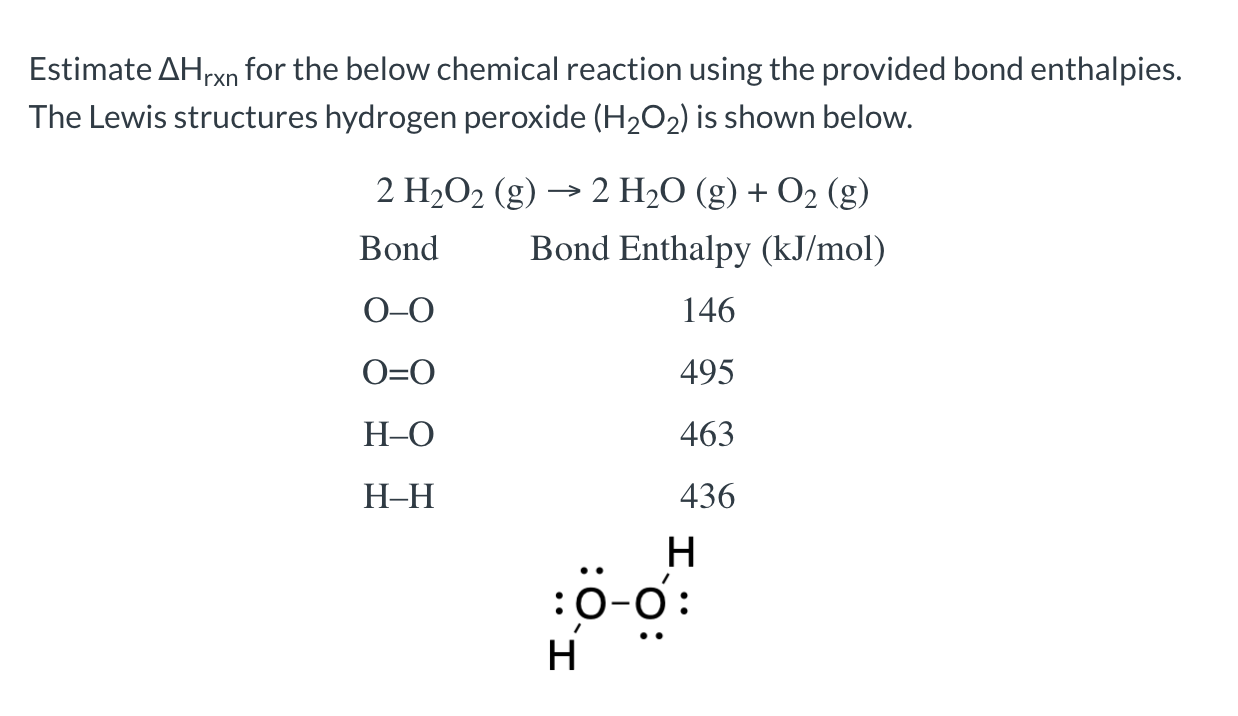


Solved Estimate Ahrxn For The Below Chemical Reaction Usi Chegg Com
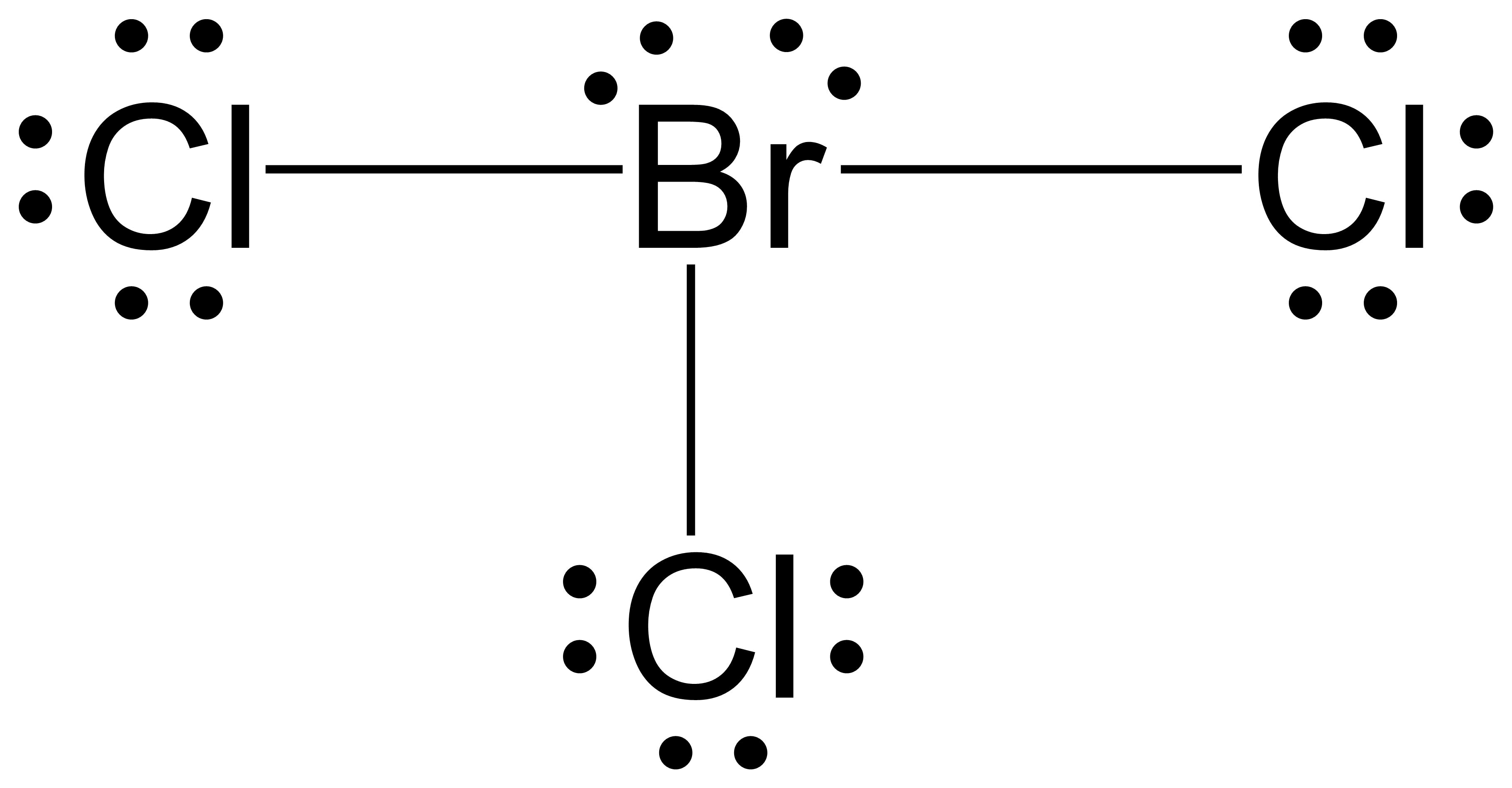


Resonance Structures And Formal Charge M8q3 Uw Madison Chemistry 103 104 Resource Book
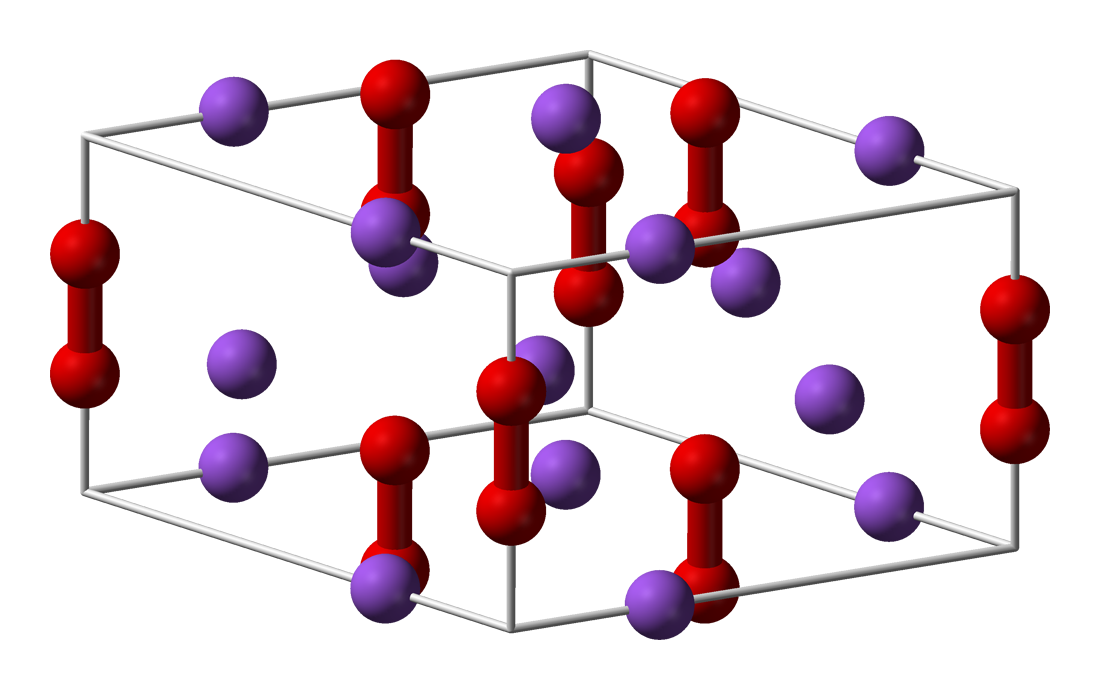


Sodium Peroxide Wikipedia



Lewis Structure Of Hydrogen Peroxide H2o2 Novocom Top
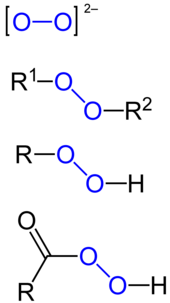


Peroxide Wikipedia
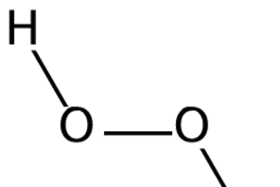


Hydrogen Peroxide Soundbite Rsc Education



0 件のコメント:
コメントを投稿